R1 Classification and structure of materials
Initial knowledge/skills rating *
September 2011
I have given myself a very low rating here just because I do not really understand what is meant by the title, but I am not going for no stars because I would at least attempt to give an answer if I was asked.
I have given myself a very low rating here just because I do not really understand what is meant by the title, but I am not going for no stars because I would at least attempt to give an answer if I was asked.
Monday 21st November 2011 **
Right with a little bit of effort here I am sure I can boost my skills rating. So to classify a material is to put it into a class, to group it. I am sure I could sort my metals, from my plastics from my woods. So what exactly is needed here? Let’s look at GCSE BBC Bite size to start with.
I have been to BBC Bite size.
I know the difference between a thermoplastic and a thermosetting plastic since doing our research for the plastics presentation. I know the difference between an alloy and a pure metal. I know a ferrous metal contains iron and a non-ferrous metal does not. Teenage brain cells refreshed over the last few weeks. I know the difference between softwood and hardwood.
I know a composite material is one containing two or more materials usually wood and a bonding agent in resistant materials. Is that right? No promolite is a composite material, so I’ll settle for two or more materials bonded together. I need to reinforce this definition?
So the only group missing for GCSE are ceramics. I had better learn about that one. A little bit of copying and pasting and attaching to my folder and I should have that one sorted too.
I seem to approach these subjects really warily as if someone is going to catch me out. It’s sort of well… yes I know this and….yes…. I know that but there has got to be more to it…… and maybe there is but not on BBC Bitesize.
So my next step to more stars is to really learn my definitions till I know them inside out. On it!
And a bit of geography wouldn't go a miss here either. So that I am clear about my tropical and temperate regions.
12th February 2012
I still need to do some work on this area. I really need to know some names of the plastics that belong to each category in fact having those at my finger tips for all categories would be a good thing. I really just need to make myself a chart to learn from.
Right with a little bit of effort here I am sure I can boost my skills rating. So to classify a material is to put it into a class, to group it. I am sure I could sort my metals, from my plastics from my woods. So what exactly is needed here? Let’s look at GCSE BBC Bite size to start with.
I have been to BBC Bite size.
I know the difference between a thermoplastic and a thermosetting plastic since doing our research for the plastics presentation. I know the difference between an alloy and a pure metal. I know a ferrous metal contains iron and a non-ferrous metal does not. Teenage brain cells refreshed over the last few weeks. I know the difference between softwood and hardwood.
I know a composite material is one containing two or more materials usually wood and a bonding agent in resistant materials. Is that right? No promolite is a composite material, so I’ll settle for two or more materials bonded together. I need to reinforce this definition?
So the only group missing for GCSE are ceramics. I had better learn about that one. A little bit of copying and pasting and attaching to my folder and I should have that one sorted too.
I seem to approach these subjects really warily as if someone is going to catch me out. It’s sort of well… yes I know this and….yes…. I know that but there has got to be more to it…… and maybe there is but not on BBC Bitesize.
So my next step to more stars is to really learn my definitions till I know them inside out. On it!
And a bit of geography wouldn't go a miss here either. So that I am clear about my tropical and temperate regions.
12th February 2012
I still need to do some work on this area. I really need to know some names of the plastics that belong to each category in fact having those at my finger tips for all categories would be a good thing. I really just need to make myself a chart to learn from.
So this is the table of information I have put together with the key facts that I want to learn
***
Plastics
Natural sources of plastics include:
Synthetic plastics are chemically manufactured from:
Plastics can be divided into two groups thermoplastics and thermoset plastics.
Thermoplastics can be heated and reformed many times. they have a memory. the long lines of molecules slip over each other allowing the plastic to take many shapes. When re heated the molecules return to their former positions and the plastic returns to its original shape. This can be easily demonstrated by heating acrylic in the oven or with a heat gun. Forming and then reapplying the heat source and watching the acrylic unfurl to its original shape. The same can be seen when using a vacuum formed and HIPS allowing more then one attempt at forming the shape. Very useful when the mould has been pressed into the plastic too soon.
Thermoset plastics can only be heated and shaped once. A bit of a clue in the name.
- plants - from which cellulose can be extracted
- trees - from which latex, amber and resin can be extracted
- animals - from which horn and milk (used to make glues) are obtained
- insects - from which shellac (used to make polish) is obtained
Synthetic plastics are chemically manufactured from:
- crude oil
- coal
- natural gas
Plastics can be divided into two groups thermoplastics and thermoset plastics.
Thermoplastics can be heated and reformed many times. they have a memory. the long lines of molecules slip over each other allowing the plastic to take many shapes. When re heated the molecules return to their former positions and the plastic returns to its original shape. This can be easily demonstrated by heating acrylic in the oven or with a heat gun. Forming and then reapplying the heat source and watching the acrylic unfurl to its original shape. The same can be seen when using a vacuum formed and HIPS allowing more then one attempt at forming the shape. Very useful when the mould has been pressed into the plastic too soon.
Thermoset plastics can only be heated and shaped once. A bit of a clue in the name.
Timber (wood)
There are two types of timber, called hardwood and softwood. These names do not refer to the properties of the wood: some softwoods can be hard and some hardwoods can be soft.
Softwoods come from coniferous trees which are evergreen, needle-leaved, cone-bearing trees, such as cedar, fir and pine.
Structure. Softwoods come from fast growing trees which have a wide grain.
Hardwood
Hardwoods come from broad-leaved, deciduous trees. The main hardwood timbers are ash, beech, birch, cherry, elm, iroko, mahogany, meranti, oak, obeche, sapele and teak.
Structure: Hardwoods are slower growing than softwoods and have a tighter grain. The appearance and characteristics differ from each species.
For example: Oak is light brown in colour with tiny straight flexs of dark brown. It is a strong, tough and heavy with an open grain used for interior woodwork and good quality furniture. It should be noted that oak corrodes steel screws and fittings. Whereas mahogany is a deep reddish brown colour. It is fairly strong, durable with some interlocking grain again used for good quality furniture.
Manmade boards are composite materials made from wood and a bonding agent , such as glue. These are Ply, MDF (Medium density fibre) and chipboard. Both chipboard and MDF are made from the waste pulp from cutting timber whereas ply wood is harvested from the timber in long thin sheets called veneers. The veneers are generally layer with the grain at right angles to the sheet above and below and joined with adhesives.
Metals
An Ore is a type of rock that contains minerals with important elements including metals. The ores are extracted through mining; these are then refined to extract the valuable element(s).
Pure metals: are rarely used in their pure form, and are usually mixed with other metals to improve their properties.
An alloy is a new metal which is formed by mixing two or more metals and sometimes other elements together. Alloys are normally grouped into ferrous and non-ferrous metals.
Ferrous: Ferrous metals contain iron. Examples are cast iron, mild steel, medium carbon steel, high carbon steel, stainless steel, high speed steel and wrought iron.
Non-ferrous : metals do not contain iron. Some common non-ferrous metals are aluminium, Duralumin, copper, zinc, brass, gilding metal,lead,zinc, silver, tungsten, titanium and tin.
Nano Technology: is the manipulation of a system at a molecular level. Our talk from Diane Asten was my introduction to this concept. I understand that by mixing metals and compounds at a molecular level it is possible to create an endless list of alloys. Diane described the process of obtaining metals with the desired properties very much in the way a chief manipulates flavours by altering ingredients.
Pure metals: are rarely used in their pure form, and are usually mixed with other metals to improve their properties.
An alloy is a new metal which is formed by mixing two or more metals and sometimes other elements together. Alloys are normally grouped into ferrous and non-ferrous metals.
Ferrous: Ferrous metals contain iron. Examples are cast iron, mild steel, medium carbon steel, high carbon steel, stainless steel, high speed steel and wrought iron.
Non-ferrous : metals do not contain iron. Some common non-ferrous metals are aluminium, Duralumin, copper, zinc, brass, gilding metal,lead,zinc, silver, tungsten, titanium and tin.
Nano Technology: is the manipulation of a system at a molecular level. Our talk from Diane Asten was my introduction to this concept. I understand that by mixing metals and compounds at a molecular level it is possible to create an endless list of alloys. Diane described the process of obtaining metals with the desired properties very much in the way a chief manipulates flavours by altering ingredients.
Ceramics
Ceramics are made from clay, sand and feldspar. These materials are ground to a fine powder, mixed together and fired at high temperatures (700-2000°C).
So what comes next?
It would be really nice to know what the composition of some of the alloys are.I remember trying to remember the composition of bronze and brass when I was at school but I invariable got hem confused. I am going to use the internet to find some answers. I am curious as to what the composition for gold and silver are.
Well the composition of brass is copper and zinc and the composition of bronze is copper and tin. I can but try and remember. Failing that I can look it up here.
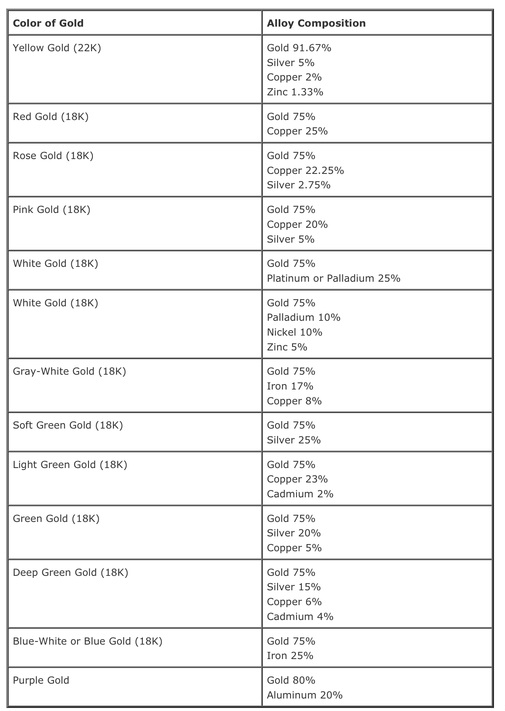
I found a table of Golds at http://goo.gl/kex8R. I never knew there were so many different coloured golds used in jewellery making. I thought you only had three yellow, white and oh the third one used in three gold rings but I do not even know what colour that is. I never knew there was such a thing as Purple gold (Gold and aluminium) Ah looking at the table it is the higher addition of copper that gives the red colours. this makes sense.Platinum and palladium give the whites. Wait a minute Gray-White gold (18k) and Blue Gold actually contain iron. This would mean that gold can be either a ferrous or non-ferrous metal depending on its grade.
Well the composition of brass is copper and zinc and the composition of bronze is copper and tin. I can but try and remember. Failing that I can look it up here.
I found a table of Golds at http://goo.gl/kex8R. I never knew there were so many different coloured golds used in jewellery making. I thought you only had three yellow, white and oh the third one used in three gold rings but I do not even know what colour that is. I never knew there was such a thing as Purple gold (Gold and aluminium) Ah looking at the table it is the higher addition of copper that gives the red colours. this makes sense.Platinum and palladium give the whites. Wait a minute Gray-White gold (18k) and Blue Gold actually contain iron. This would mean that gold can be either a ferrous or non-ferrous metal depending on its grade.
It really is so easy to use the internet for research. I was able to look up the composition of my favourite metals (I just like their names) like titanium, tungsten and aluminium. What I discovered was that they often have long lists of possible elements that would not be easy to remember.I do not feel it is essential to post my findings here but to make a note to search "Composition of..X...in Goggle" will give you a wealth of answers. The other important fact I discovered was that the grades of metals could lead them into either category of ferrous or non- ferrous. Where I had previously typed in is " Is X a ferrous metal" my results were general but when I researched more deeply I discovered that the grades of aluminium, gold and titanium mean it could actually on occasions fall into the ferrous category. For example aluminium may not contain iron OR it could contain up to 0.4% iron. This may not be a lot but if it is enough to change the characteristics of a metal then it counts.
Stainless Steel
The one last table I want to add here is the table I found for various grades of stainless steel. I want to include this table because I came from an industry that used steel so it is an area I have some familiarity and understanding for. What I liked about this table was the explanation of what different metals were doing to the characteristics of steel. It really is like a recipe book for metals.
Skill ratings ***
Although I feel my knowledge is sound for this subject I feel I will only feel properly confident when I start sharing it out loud. This could be something I will start subjecting my family to. However I am sure I will be both confident and competent with a little practise.
Work for another day
Whilst looking for the composition of various metals I occasionally found them written in symbols as used in the periodic table. This makes me realise the link between science and the atomic structure of metals. This is something I will have to leave for another day but becoming more familiar with the periodic table and its uses is certainly something I would like to do in the future.